Two-Thirds of Survey Respondents Plan to Invest in Single-use Bioreactors
According to survey responses exclusive to IBO, use of single-use bioreactors is growing due to increased bioprocessing activity. Bioprocessing is rapidly expanding as the global demand for biomolecule-based therapeutics increases. Bioprocessing is a unique production method that utilizes living cells to produce biomolecules of interest. Cells are grown in bioreactors, which are vessels that are integrated with complex control and monitoring systems to create an environment that supports cellular growth. This unique production method, coupled with the stringent manufacturing requirements for pharmaceutical products, makes contamination a key concern in bioprocessing. Single-use bioprocessing equipment mitigates contamination risk, as the components that contact cell cultures arrive sterile and are discarded after each use.
Single-use bioreactors are equipped with a sterile disposable bag, pouch or rigid plastic vessel in which cells are grown. They offer several advantages over conventional, reusable bioreactor systems in terms of reducing contamination risk, faster turnover between cell batches, fewer maintenance and cleaning requirements, and overall reduction of labor time.
However, single-use models have certain limitations compared to their conventional counterparts, such as difficulties in scale-up and scale-down, non-customizability, lower rates of oxygen transfer, challenges with efficient mixing and agitation, and disposal issues. While conventional bioreactors can be customized for volumes of 100,000 L or more, single-use bioreactors are generally only available in volumes up to 2,000 L. Despite these trade-offs, many bioprocess users are converting to single-use bioreactors.
Two hundred and twelve users of bioreactors were surveyed in July as part of an SDi report on bioprocessing technologies. This report includes detailed market data, analysis and end-user perspectives for key upstream and downstream bioprocess instruments and consumables.
Respondents Plan New and Additional System Purchases
To gauge expected purchases, respondents were asked about their plans regarding single-use bioreactors within the next two years. Those who do not currently use single-use systems were asked whether they plan to invest in adopting the technology, while respondents who already utilize single-use bioreactors were asked whether they plan to expand their use.
Of the respondents who were not current users of single-use bioreactors, over two-thirds indicated that they were planning on investing in these systems. These investments in single-use bioreactors may represent either the replacement of conventional systems, or expansion of capacity for use in addition to existing conventional systems. Respondents who already utilize single-use bioreactors were very likely to purchase additional systems, as 80% reported that they expect to expand their use of single-use bioreactors in the next two years.
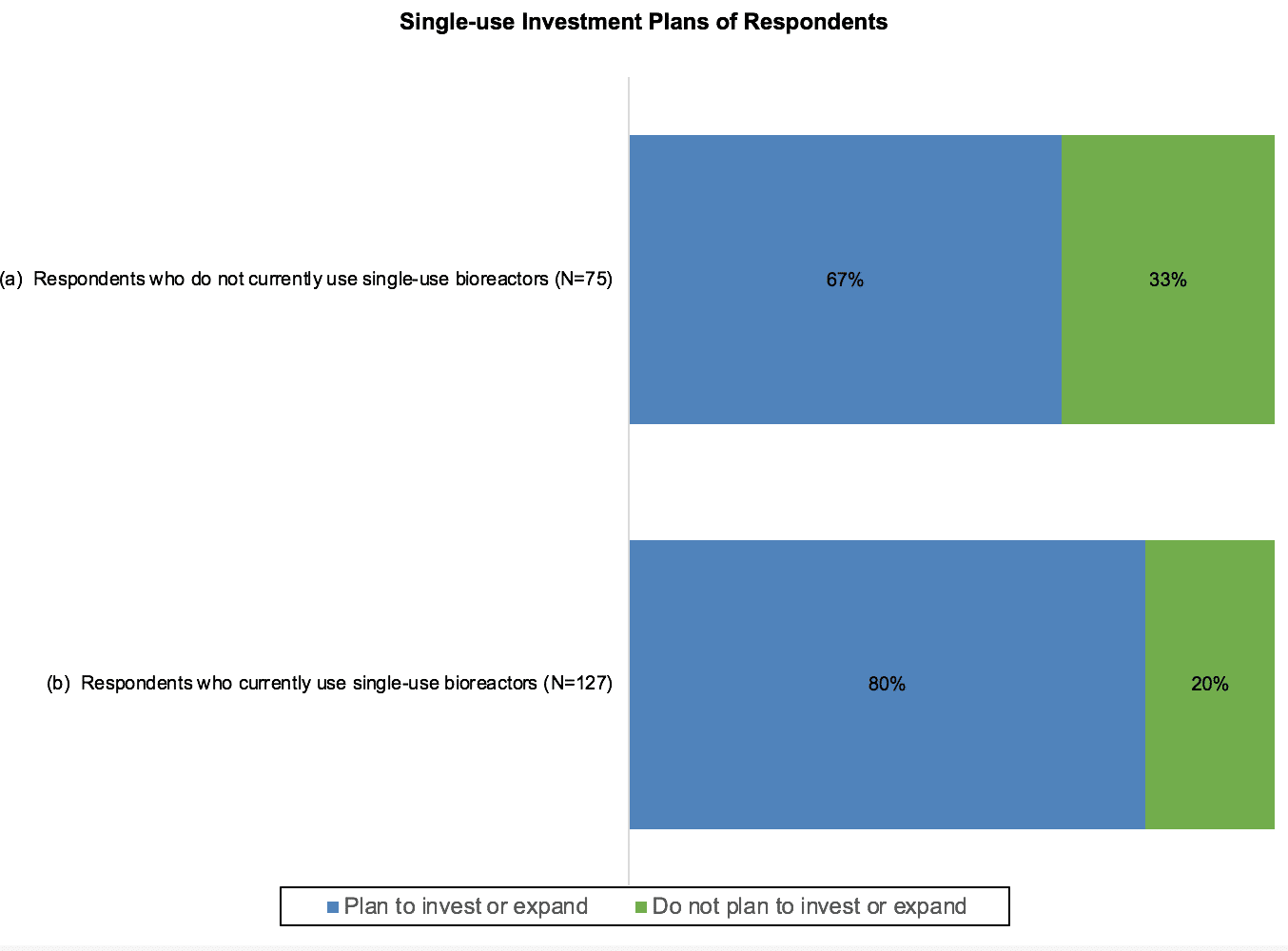
Click to enlarge
High Demand for Basic Research
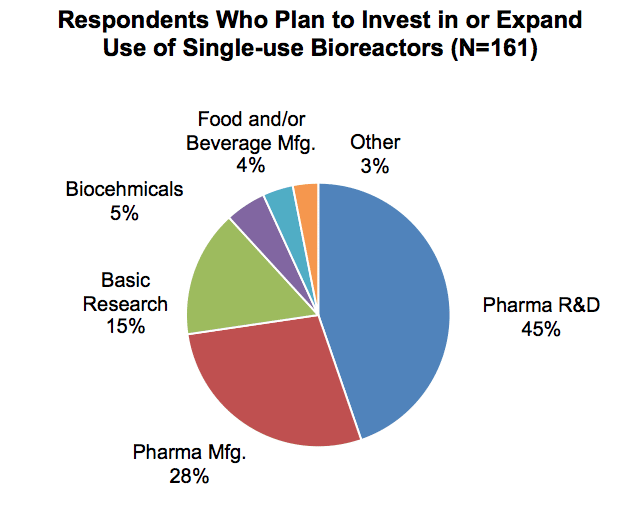
Respondents who expect to purchase single-use bioreactors in the next two years were predominantly from the pharmaceutical field. Nearly half participated in pharmaceutical R&D, while over a quarter were involved in the manufacturing side. The pharmaceutical and biotechnology sector is the largest consumer of bioprocessing technologies in general, but demand for single-use technologies is particularly strong given the stringent purity required of therapeutic products.
Basic research was identified as a primary function mainly by respondents from the public sector, though some pharma and biotech, and agriculture and food sector respondents also selected this as their organization’s primary function. Participants in basic research accounted for 15% of respondents who indicated that they expected to purchase single-use bioreactors in the next two years.
Click to enlarge
Pilot-scale Bioreactors Most in Demand
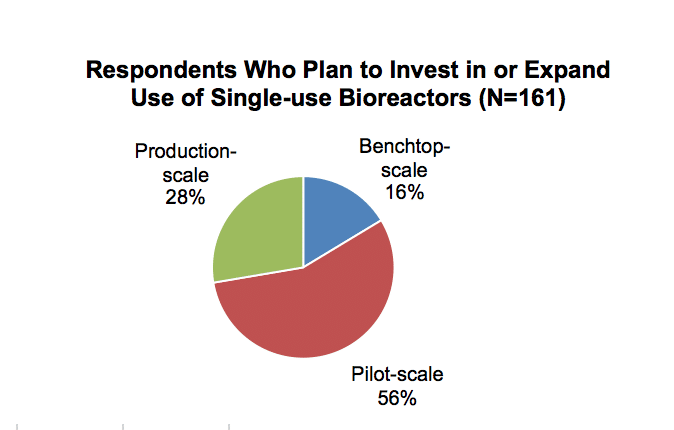
Bioreactors can be broadly categorized into three capacity ranges: benchtop scale, pilot scale and production scale. While there is much variation in how suppliers and users define these scales, for the purposes of the bioprocessing technologies report, SDi has defined benchtop-scale as up to 10 L, pilot-scale is 10–100 L and production-scale as greater than 100 L. Pilot-scale single-use bioreactors are the most common capacity range used by respondents. More than half of respondents who expect to make single-use bioreactor system purchases in the next two years use this size range. Pilot-scale bioreactors are suitable for both R&D and smaller-scale production.
Single-use bioreactors are quickly gaining market share in all regions. Demand for single use is expected to exceed that of conventional bioreactors by 2022. Biologics-based therapeutics represent a growing portion of pharmaceutical R&D pipelines, as these products increasingly gain regulatory acceptance and consumer demand grows.
Click to enlarge

