A Big Year for Illumina
As part of its 2019 Annual Forecast Issue, IBO named Illumina our company of the year for 2018. The company’s achievements included its strongest revenue growth since 2015, the adoption of its NovaSeq system by new users, and advancing the use of NGS in the pharmaceutical and diagnostics end-markets. Specifically, the year was marked by sales growth propelled by the Genomics England population genomics program, arrays for direct-to-consumer testing and NovaSeq consumables.
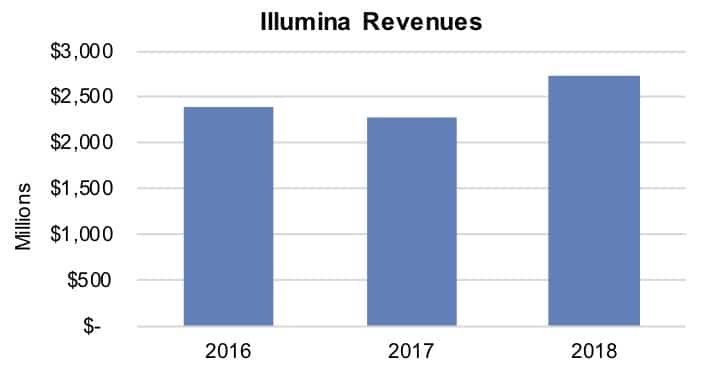
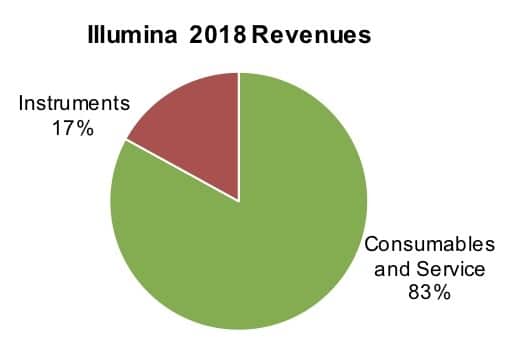
The company announced its year-end results on January 29 (see Bottom Line). “Illumina delivered its 20th year of sequential revenue growth in 2018, with revenue of approximately $3.3 billion, up 21% from 2017,” said company President and CEO Francis deSouza and Eric Endicott, senior director, Corporate Communications. Notably, the growth reflected robust sales across product lines. “Sequencing consumables grew about 23%. Sequencing systems grew about 10% and microarrays grew about 22%,” they noted.
Adoption of the NovaSeq sequencer was one contributor to this growth, reflecting the ability of the company to broaden the use of whole-genome sequencing through the system’s introduction. “Customers are embracing this power to sequence larger cohorts more broadly and deeply,” they said. “Since its launch 2 years ago, NovaSeq users have sequenced more than 600,000 whole-genome equivalents. That’s 3 times as many as were sequenced on the HiSeq X in its first 2 years of launch.” The HiSeqX was previously Illumina’s highest-throughput sequencing system with an output per run of 1.8 Tb, compared to 1–6 Tb for the NovaSeq depending on the flow cell (using two flow cells per run).
Through both the replacement cycle and new users, NovaSeq’s is expected to also grow instrument and consumables revenue going forward, fulfilling a strategic goal of Illumina’s for last year. “We placed more NovaSeqs in 2018 than we did in 2017, including more than 100 units in the fourth quarter alone, a record. To date, new-to-Illumina or benchtop conversions represent approximately 30% of our NovaSeq installed base, and three-fourths of our HiSeq customers have yet to purchase a NovaSeq System,” they said. “This group represents approximately 630 HiSeq customers, supporting our confidence in a steady, multiyear transition.”
Illumina has pointed out over the years its role in developing the NGS market as a whole. This role can be attributed to the company’s market share, technology and resources. In particular, these efforts have been aimed at applications beyond the research lab. The company cited several accomplishments in 2018 in this regard in the areas of NIPT, RUGD (rare and undiagnostic genetic disease) and oncology testing, marking the maturing of these markets. “In NIPT, our VeriSeq CE-IVD customers ordered over 300,000 tests in 2018, up more than 70% from the previous year, highlighting the rapidly growing clinical NIPT opportunity,” they said.
Greater reimbursement opportunities are among the most important signals of success in increasing clinical and diagnostic opportunities. “Reimbursement has made rapid progress in the last 2 years, with more than 147 million patient lives in the US now covered for RUGD whole-exome sequencing,” explained Mr. deSouza and Mr. Endicott. “Last week, CMS’s [Center for Medicare & Medicaid Services’] final CPT code came into effect, pricing whole-genome sequencing at more than $5,000 for a single genome for RUGD patients. This is a critical benchmark for whole-genome sequencing in the Medicare and Medicaid populations.”
Further detailing these numbers, Mr. deSouza and Mr. Endicott commented, “We’re seeing significant progress in reimbursement. In 2018, 50 million additional U.S. lives gained coverage for NGS panels, bringing the total to over 200 million lives.” Additionally, in 2018, regulatory and professional recognition of the NGS’ utility increased. “In 2018, 5 large gene panels received breakthrough designations or approvals for therapy selection and minimal residual disease assessment, up from 1 in 2017. NCCN [National Clinical Practice Guidelines in Oncology] and ESMO [European Society for Medical Oncology] guidelines now include NGS testing for several types of cancer, including TMB assessment in lung cancer,” they explained.
In oncology, NGS’ largest market, the company also reported wider NGS adoption, particularly among pharmaceutical companies. “In 2018, we saw significant progress in oncology research to support basic discovery and therapy development, as well as regulatory approvals and reimbursement of oncology NGS tests that will drive increasing adoption of sequencing as a standard of care in cancer treatment,” they said. In particular, they explained, adoption was strong for use in clinical trials. “For example, NGS is included in many of the 2,250 active clinical trials in immuno-oncology registered in the US, driven by the emergence of genomic biomarkers like tumor mutational burden or TMB.”
Among Illumina’s larger investments during the year were the purchase of two companies. One acquisition, Edico Genomics (see IBO 5/15/19), was a step up in the company’s exposure to the software market for variant analysis. Edico’s DRAGEN platform provides a hardware and software solution for secondary analysis of NGS data. “As sequencing is getting more adopted in the clinic/standard of care, we want to move the focus from raw data to analyzed/variant calls. Doing so allows customers to focus on higher-impact work interpretation and actionability of information,” explained Mr. deSouza and Mr. Endicott. “By combining DRAGEN with our technologies, we’re taking data analysis from a complicated and costly process to a push-button result. The acquisition supports our informatics strategy is to provide the infrastructure that gets customers up and running—from sequencing to result—in the easiest and most cost-effective way possible.”
The other major purchase was of Pacific Biosciences (see IBO 11/15/18), giving Illumina a presence in the long-read market, and providing another path for future sales growth with new sequencing capabilities. As Mr. deSouza said on a November 2018 conference call discussing the deal, “Pacific Biosciences’ accurate native long-reads averaging 15 to 30 kilobases provide valuable insights around long-range rearrangements, structural variants and haplotypes, which can be challenging using short-read technologies.” In a January SEC Proxy filing, Pacific Biosciences estimated 2019 revenues of $144 million, up 6.2%, and an improvement in operating income from a loss of $83 million to a loss of $48 million, with an operating profit expected by 2020.
With these acquisitions representing major strategic activities and future prospects, and a meaningful acceleration in revenue growth and profitability as well as market access, 2018 was a big year for Illumina.